SUMMARY
Some embodiments of the present invention are generally directed to providing an isolated antiviral peptide and a significant in vitro binding affinity for HIV-1 gp 120, gp 41 and human CD4 cells. The peptide has anti-retroviral activity in vivo, particularly anti-HIV-1 activity. The peptide, referred to herein as IPF (Inactivated Pepsinogen Fragment), was isolated from porcine pepsinogen, purified, and irreversibly inactivated for use in HIV-1 prophylactic, therapeutic and diagnostic procedures.
Other embodiments of the present invention are generally directed to providing pharmaceutical compositions comprising IPF and methods for preventing, treating, and diagnosing HIV-1 infections and HIV-1 related conditions such as AIDS (Acquired Immune Deficiency Syndrome) and ARC (AIDS Related Complex) with these compositions.
DETAILED DESCRIPTION OF THE INVENTION
Pepsins (of which there are several isozymes) are the principal proteases in gastric secretions of adult mammals. They belong to the family of aspartic proteases and are synthesized and secreted by cells in the gastric mucosa as inactive enzyme-precursors consisting of a signal peptide, an activation peptide and an occluded active enzyme. En route to the lumen of the stomach for protein digestion, the signal peptide is cleaved to yield the inactive proenzyme pepsinogen, which, on exposure to a low gastric pH (<4), cleaves in turn to yield mature, catalytically active pepsin.
Porcine pepsin was one of the first enzymes to be studied, and is perhaps the best-understood aspartic protease. It has 327 amino acid (aa) residues, and a molecular mass of 34kDa (PNAS (U.S.) 70:3437-39 1973). Proteolytic activity of pepsin is at its highest at a pH of about 1.8 to 3.5; it is inactivated at a pH of about 5 and irreversibly inactivated (denatured) at a pH of about 6-7. Owing to their importance, amino acid residues associated with the substrate binding (active) site have been a research focal point. However, it is apparently still not clear how much functional activity, if any, is influenced by the remainder of the peptide.
The family of aspartic proteases (aspartases) is characterized by aspartic acid residues at their active (catalytic) sites. Human pepsin, for example, has two active site aspartate residues (coded "D" or "Asp"). This family also includes the HIV protease (and its numerous variants), comprising two identical chains each having a single active-site aspartate residue. Essential for maturation of the newly synthesized virus, which is expressed as a polyprotein, this protease has become a popular target for researchers attempting to block HIV replication.

Homologues or analogues of the sequence which conserve at least critical binding site amino acid structures and functions and also conserve any distal structural/functional residues essential for binding activity, as described herein, may be substituted for the IPF of SEQ ID:NO 1. Variants of the sequence, including chemically modified derivatives, having a high sequence similarity will be generally preferred, provided that binding activity is not significantly adversely affected. Residues superfluous to the disclosed function of the peptide of the invention may be deleted or added with the same proviso. Modified sequences may be evaluated for conserved binding activity by, for example, following the binding assays described herein or in the literature. Numerous databanks are accessible for protein sequence analysis, such as those which combine sequence similarity with fold recognition to predict functional equivalents. Binding properties (affinity, specificity, etc.) may also be evaluated by the binding assays described below or other conventional assays, as known in the art.
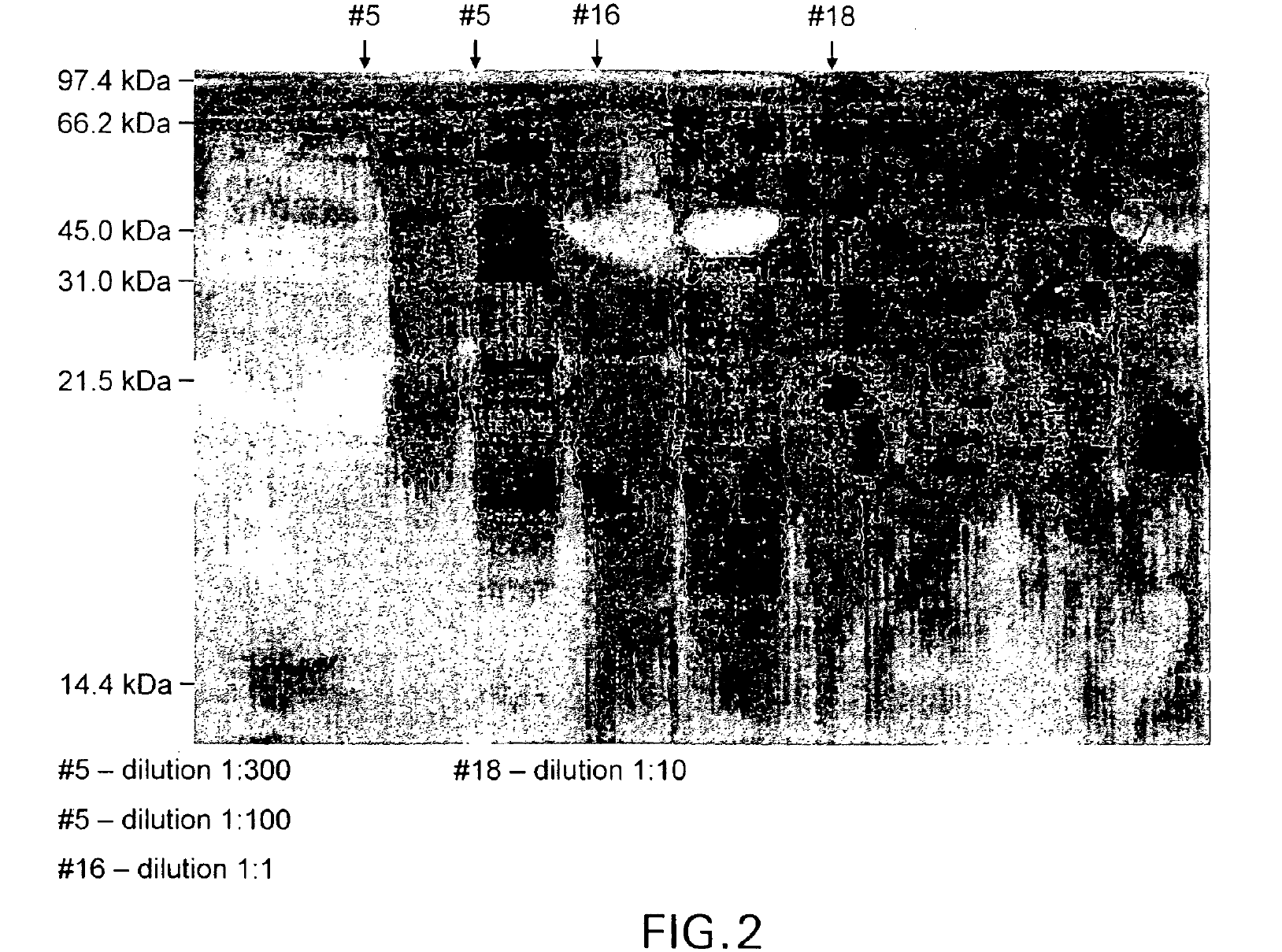
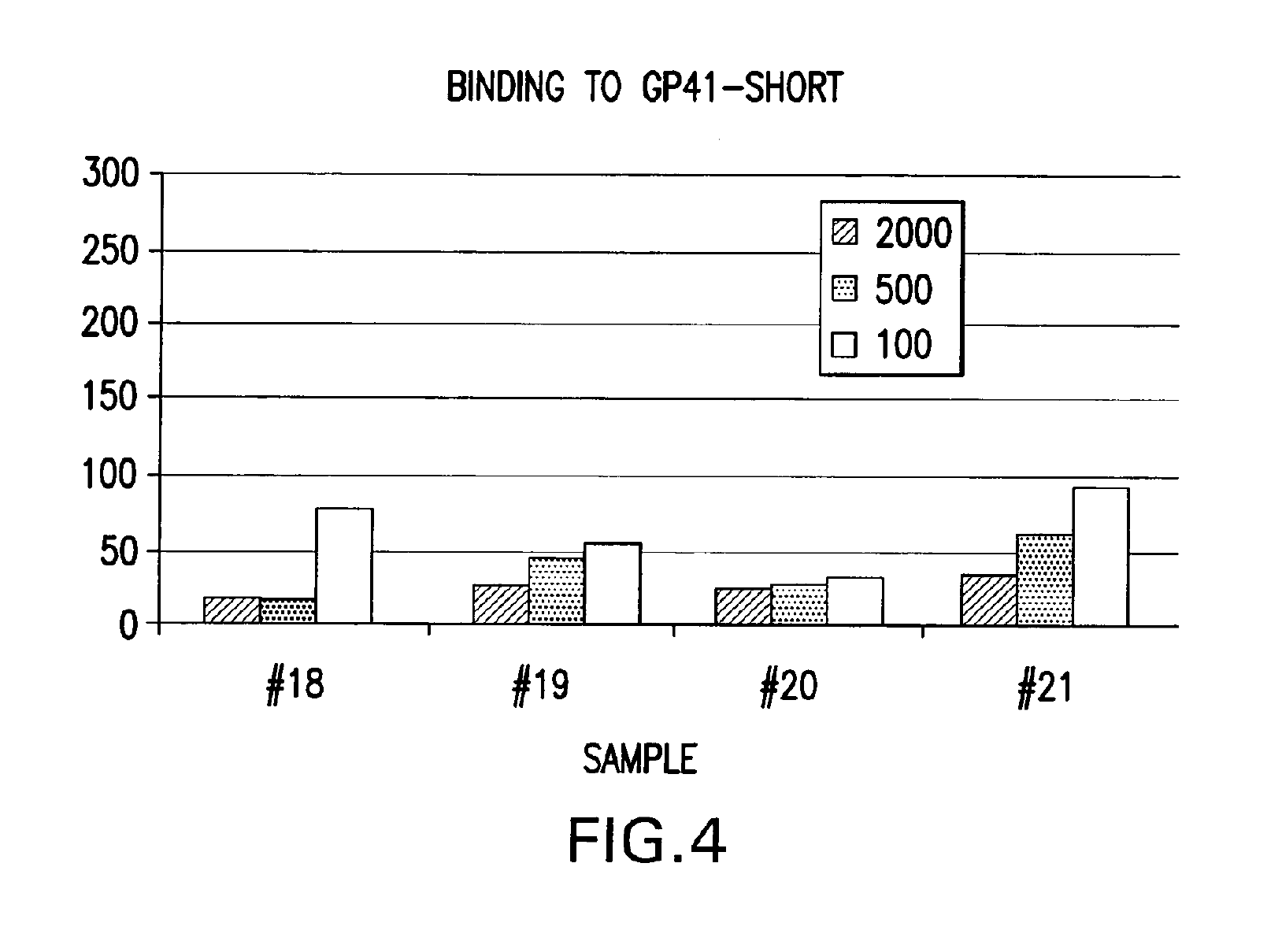
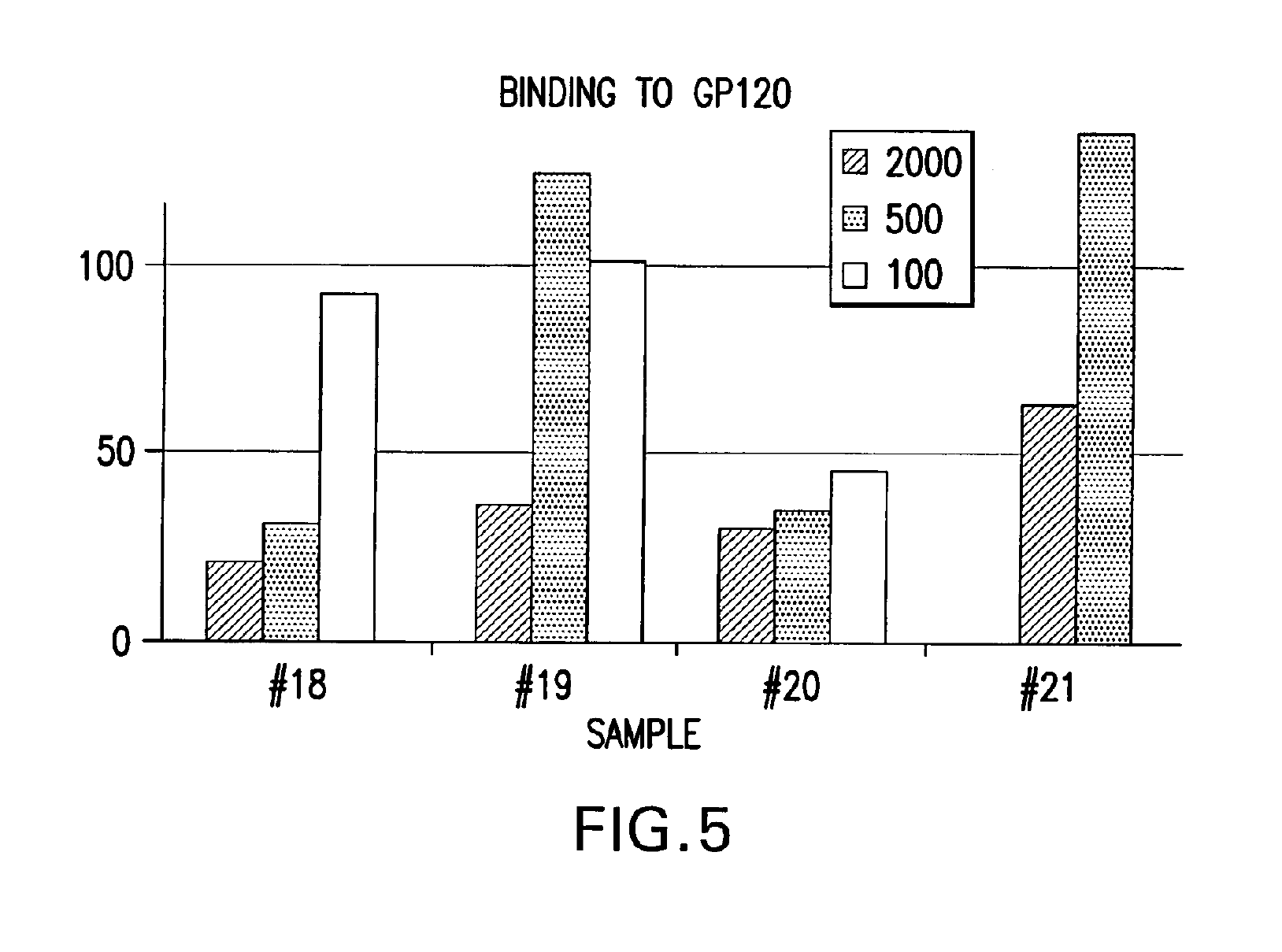
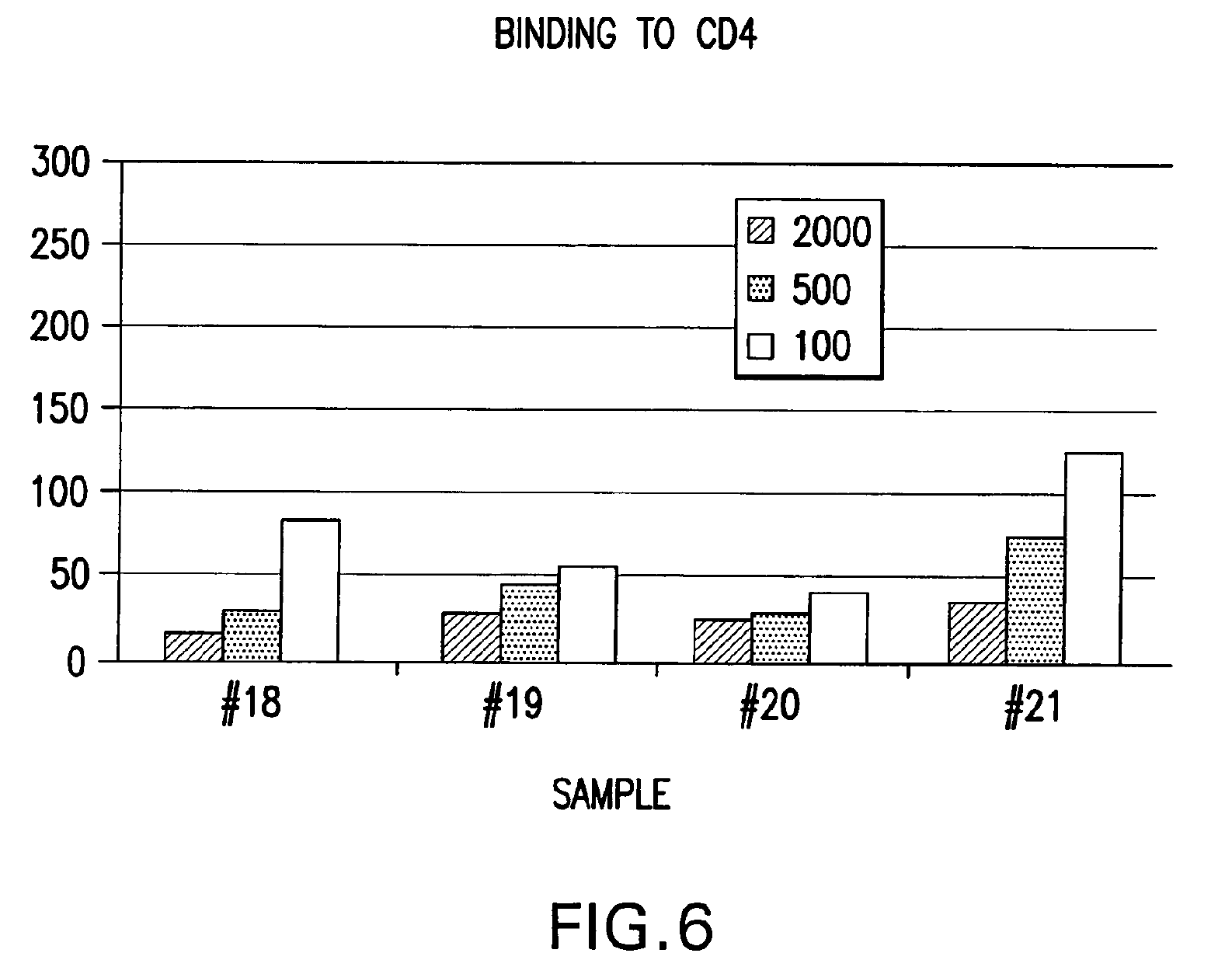
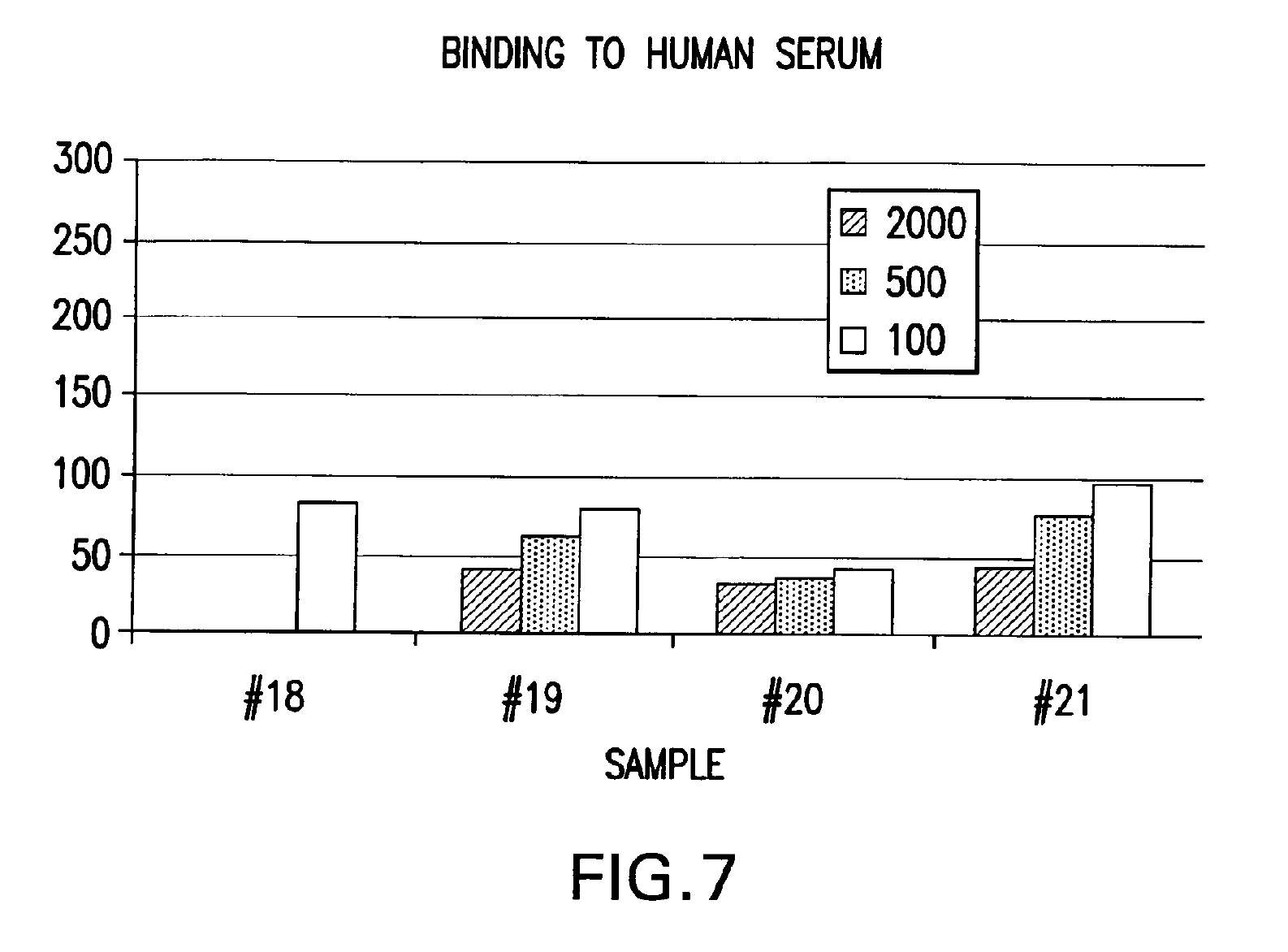
IPF demonstrates binding in vitro with nonglycolysed fragment 579-601 of the HIV-1 envelope protein gp41 subunit (FIGS. 4 and 8), with gp120 HIV-1 subunit (FIG. 5), with human CD4+ cells (FIG. 6), and with human serum (FIG. 7) under gel electrophoresis. The spontaneous binding of IPF with the gp41 subunit is a particularly important biological property. Separately, under simple agarose electrophoresis, IPF and gp41 move in opposite directions. However, when they are mixed prior to electrophoresis, gp41 changes direction and takes the direction of IPF. Quantitative analysis showed that the binding capacity ratio of IPF to gp41 was 1:0.66. That is, three molecules of IPF bound two molecules of gp41 to form a complex which may function in vivo as, for example, a superantigen with significant anti-HIV-1 biological activity. Such antigen can be used as a bioassay reagent, in the production of mono- or polyclonal antibodies, in the manufacture of vaccines, and in other applications wherein antigens are conventionally employed. While the mechanism of these binding events is not completely understood at this time, it is contemplated that exposure of HIV-1 to the IPF of the present invention will effectively block gp41 and gp120 domains essential for viral entry into CD4+ cells and inhibit viral infection, in vivo and in vitro. It is also contemplated that the IPF of the present invention will effectively compete with HIV-1 for its CD4+ cell surface binding sites and inhibit virus entry into these cells, in vivo and in vitro. Various in vitro protocols are known in the art for predicting in vivo antiviral activity of compounds for inhibiting replication of HIV, and these protocols may be used in connection with the practice of the present invention. Exemplary art-recognized anti-HIV screening assays are cited in U.S. Pat. No. 5,869,522, issued 9 Feb. 1999 to Boyd et al., including those described in J.Virol.Methods, 33:87-100,1991; J.Natl.Cancer Inst., 81:577-586, 1992; and J.Med.Chem. 35:1978-1986, 1992, and Boyd, M. R., in AIDS Etiology: Diagnosis, Treatment, and Prevention, pp305-319 (Lippincott, 1988, DeVita, V. T., Jr., et al., eds). In accordance with one aspect of the present invention, IPF is used to diagnose viral infection, particularly HIV-1 infection. Bioassays suitable for this purpose are well-known and routine. Typical of these are assays based on competitive binding between, for example, a known amount of a viral protein and a biological sample to be tested for the same viral protein, using an excess of antiviral reagent capable of specifically binding with the known protein, such as an antibody. A mixture of these is incubated and the amount of bound complex calculated and compared to that in a control mixture lacking the sample. The presence, if any, and amount of the viral protein in the sample can then be determined. There are numerous variations on this process, such as sandwich assays, assays with immobilized reagent, assays using labeled reagent (e.g., ELISA, RIA, FIA), and so on. Whatever the variation, whether for detecting or quantifying complex, or for additional reagents, or other modification, they all require a binding agent for the unknown sample. Any of these routine binding assays for quantifying or identifying an unknown sample may thus be used in the practice of the present invention by substituting IPF as the antiviral binding agent for samples to be tested for HIV-1 gp120, gp41, or infected CD4+T- cells.

In accordance with another aspect of the present invention, IPF is used as a prophylactic or therapeutic to prevent or to treat HIV infections. (Herein the term "HIV infections" refers to AIDS and ARC in addition to viral infection per se unless otherwise noted). For in vivo use, IPF may be prepared for administration by mixing it at the desired degree of purity with a pharmaceutically-acceptable carrier suitable for the route of administration, as well-known in the art. Although IPF is desirably administered with an adjuvant in some applications, in situations where a series of IPF doses are administered, boosters with IPF may not require adjuvant. Intramuscular or subcutaneous injections are presently the contemplated route for both therapeutic and prophylactic administration of IPF. However, intravenous delivery, delivery via catheter or other surgical tubing, or other parenteral route may also be used. Alternative routes include oral routes for administering tablets, liquid formulations and the like, as well as inhalation routes. Liquid formulations reconstituted from powder formulations may be utilized. IPF may also be administered via microspheres, liposomes, or other microparticulates, and via delivery systems or sustained release formulations dispersed in certain tissues including blood.
The dosage of IPF administered will depend upon the properties of the formulation employed, e.g., its binding activity and in vivo plasma half-life, the concentration of IPF in the formulation, the administration route, the site and rate of dosage, the clinical tolerance of the patient involved, the patient's condition, and other considerations, as known in the art. Different dosages may be utilized during a series of sequential treatments. The practitioner may administer an initial dose and then boost with relatively smaller doses of IPF. The dosages of IPF may be combined with other HIV antivirals, such as AZT.
The following is an example of a contemplated IPF formulation, dosage and administration schedule:
The patient is administered an intramuscular injection containing 8 mg of IPF (preferably 2 ml of a formulation containing 6 mg/ral of IPF in a pharmaceutically acceptable solution) or 57 .mu.g of IPF protein per kg body weight of the patient. Each treatment course consists of 16 injections, with two injections on consecutive days per week for 8 weeks. Three months after the last injection, if the patient's condition warrants, the treatment regimen is repeated. The treatment regimen may be repeated until satisfactory results are obtained, e.g., a halt or delay in the progress of the infection or disease, an alleviation of the infection or disease, or a cure is obtained. Preferably, in this application, IPF will be formulated with an aluminum hydroxide (Al(OH)3) adjuvant. Aluminum hydroxide is a widely used adjuvant, especially in commercial products such as vaccines. It is well suited for strong antigens. Many sources of aluminum hydroxide are available. The adjuvant is commercially available under the trade name Alhydrogel.RTM. by Accurate Chemical & Scientific Co. of Westbury, N.Y. In one example, the final 1 ml of the final IPF formulation may contain: 4 mg IPF (purity > 96% .+-.0.290); 2.26 mg 0.016M AlPO.sub.4 (or 0.5 mg A1.sup.+3); 4.1 mg 0.004M CH.sub.3COONa; and 12.9 mg C.sub.6H.sub.5O.sub.7 (sodium citrate); pH 6.2. In one regimen, 2 ml of this formulation makes up one vial with the dosage per patient per day being 16 vials. During the regimen, the patient should be monitored to assess the effectiveness of the regimen. CD+4 cell counts are useful and common methodology for evaluating HIV infection, as are assays for antibody or T-cell titers.
Immunotech IPF
· Unique, patent-protected HIV/AIDS therapy
· Turns on the immune system to fight HIV infections ― not achieved with other therapies
· Inhibits the infection of CD4 T-cells by HIV
· Raises CD4 T-cell counts to healthier levels
· Reduces HIV viral loads
Two Modes of Action
1. Directly prevents HIV from infecting CD4 T -cells
2. Activates the Th1 immune response, i.e., turns on macrophages that actually ingest HIV itself, killer Tlymphocytes that destroy HIV-infected cells and other HIV-fighting immune system responses.
Only HIV/AIDS therapy to achieve this
Mode 1
By binding to specific proteins on the outer surface of HIV particles, Immunotech IPF directly prevents HIV from infecting CD4 T -cells. HIV gp120 binds to CD4 on T-cells and then to a coreceptor.
Causes gp41 attachment to the cell membrane = virus -cell fusion and HIV infection.
IPF inactivates gp120 and gp41 = no HIV binding to cells and no HIV infection.
Mode 2
Immunotech IPF binds to T-cells and turns on Th1 immune system responses that kill HIV and HIV infected cells.
Not done by any other HIV/AIDS therapies. IFN-γ, IL-2 and TNF-a are cytokines, small proteins released by T-cells and received by other cells to generate a specific immune response.
Current Antiretroviral Therapies Have Serious Limitations
· HIV infections most treatable during its earlier stages
· But cannot take antiretrovirals during earlier stages, since drug resistance so often develops.
· Which means limited or no treatment options when viral load and CD4 cell counts are at their worst, i.e., AIDS.
· Non-antiretroviral treatments like Immunotech IPF remain lacking but are in tremendous demand.
Immunotech IPF Advantages
•Replaces or complements current antiretroviral therapies
•Potentially lesser costly and much less toxic
•May be effective as a periodic therapy instead of a daily one
•Likely unaffected by HIV mutations that can hamper antiretroviral therapies (HAART).



